Ultimate Guide to Protein Bioavailability

Ultimate Guide to Protein Bioavailability
Protein bioavailability tells you how much of the protein you eat your body can actually absorb and use. It’s not just about the grams listed on a label but how efficiently your body converts that protein into amino acids for muscle repair, immune function, and tissue maintenance. For example, whey protein (DIAAS score: 107%) is more usable than pea protein (DIAAS score: 62%).
Here’s what impacts protein absorption and how to make the most of it:
- Protein Type: Animal proteins (milk, eggs, meat) are generally absorbed better than plant proteins. Combining plant proteins (e.g., rice + beans) can improve their amino acid profile.
- Preparation Matters: Cooking can improve protein absorption, but overcooking can reduce amino acid availability.
- Dietary Pairing: Eating foods with anti-nutrients (like fiber or tannins) can block absorption, but soaking, sprouting, or fermenting plant foods helps.
- Individual Factors: Age, gut health, and timing (e.g., post-workout protein intake) also play a role.
Quick Tip: Focus on high-DIAAS proteins like milk, eggs, or whey for better absorption. If you’re plant-based, combine proteins and use preparation techniques to improve quality.
Keep reading for a detailed breakdown of how to choose, prepare, and time your protein intake for optimal results.
Protein Digestion & Absorption
sbb-itb-a09e3e8
What Is Protein Bioavailability and Why It Matters
Protein bioavailability is all about how efficiently your body can digest, absorb, and use the protein you eat. It’s not just about the number of grams listed on a label - it’s about how much of that protein is actually converted into amino acids that your body can put to work. These amino acids are crucial for things like muscle repair, hormone production, immune support, and maintaining healthy tissues.
Here’s how it works: Protein is absorbed in the small intestine, with the process wrapping up in the terminal ileum. As Suzanne M. Hodgkinson from Massey University explains:
Absorption of amino acids finishes at the end of the small intestine; the terminal ileum.
Once protein reaches the large intestine, it’s no longer usable for muscle-building. Instead, it gets fermented by gut bacteria. This makes the efficiency of digestion and absorption in the small intestine a key factor in how beneficial a protein source is.
For muscle building and recovery, high-bioavailability proteins are especially important. They deliver essential amino acids - particularly leucine - in amounts sufficient to kickstart muscle protein synthesis. Older adults, who naturally experience muscle loss with age, may benefit from consuming 1.2 to 1.6 grams of protein per kilogram of body weight from easily digestible sources to help maintain muscle mass.
Bioavailability also matters for weight management. Eating protein that your body can effectively absorb and use is far more beneficial than consuming protein that mostly passes through unabsorbed. While the Recommended Dietary Allowance (RDA) for healthy adults is 0.83 grams of protein per kilogram of body weight per day, this assumes the protein source is high-quality and easily absorbed.
The bottom line? Two foods might have the same protein content on their labels, but their impact on your body can vary widely depending on bioavailability. Understanding this difference can help you make smarter choices when selecting your protein sources.
Factors That Affect Protein Bioavailability
When it comes to protein, not all sources are processed by your body in the same way. Several factors influence how efficiently your body absorbs and uses the protein you consume. Essentially, bioavailability determines how much of the protein you eat is converted into usable amino acids, and several key elements play a role in this process.
Protein Digestibility
Digestibility is the cornerstone of protein bioavailability. Suzanne M. Hodgkinson from the Riddet Institute explains:
Amino acid digestibility, the disappearance of the amino acids from the gut following consumption of the protein source, is measured to determine amino acid availability.
The most accurate way to measure digestibility is at the terminal ileum (the last part of the small intestine). Older methods, like measuring protein in feces, are less reliable. This is because more than 80% of amino acids found in feces, especially in pigs, come from gut bacteria rather than the protein consumed. For example, glycine shows an ileal digestibility of 0.72 but a fecal digestibility of 0.87 - a 15% difference that could overstate protein absorption.
Most food proteins have ileal digestibility values above 80%, but individual amino acids within the same source can vary widely. In black beans, for instance, cysteine has a digestibility of just 0.302, while reactive lysine reaches 0.829.
Amino Acid Profile and Composition
For your body to effectively build and repair tissues, it needs all nine essential amino acids (EAAs). If even one of these is missing, the body’s ability to use the protein is limited by the least available amino acid. Among these, leucine plays a particularly important role, as it directly stimulates muscle protein synthesis and helps regulate energy balance.
Animal proteins typically provide all nine EAAs in sufficient amounts. On the other hand, plant proteins often lack one or more. For example, rice protein is low in lysine, while pea and soy proteins are low in methionine. To address this, combining plant proteins - like a 70:30 blend of pea and rice protein - can create a more complete amino acid profile, making the protein more effective.
Processing and Preparation Methods
How you prepare protein-rich foods can significantly influence their absorption. Simon M. Loveday from the Singapore Institute of Food and Biotechnology Innovation highlights:
The rates of dietary protein digestion and absorption can be significantly increased or decreased by food processing treatments such as heating, gelling and enzymatic hydrolysis, with subsequent metabolic impacts.
Moderate heating can improve protein digestibility by inactivating enzyme inhibitors in foods like legumes and denaturing proteins, making them easier for digestive enzymes to break down. However, extreme heating - especially when sugars are present - can trigger the Maillard reaction. While this process enhances flavor by browning foods, it can also reduce lysine availability. For example, milk proteins with 20% glycation reduce plasma lysine by 35%, and 50% glycation results in a 92% reduction.
Hydrolyzed proteins, which are pre-broken into smaller peptides, are absorbed more quickly than intact proteins. Even simple preparation changes can make a difference. For instance, stirring gelled foods like yogurt breaks up protein aggregates, speeding up digestion. One study found that peak serum amino acid levels were 13% higher after consuming stirred yogurt compared to UHT milk.
Interactions With Other Dietary Components
The foods you eat alongside protein can impact its absorption. For instance, high dietary fiber thickens digestive contents, potentially causing some protein to pass through unabsorbed. Additionally, anti-nutrients like trypsin inhibitors and tannins - commonly found in plant-based foods - can interfere with digestive enzymes and reduce protein absorption.
Fortunately, many anti-nutrients can be neutralized through preparation methods like soaking, sprouting, and cooking. Fermentation is another effective technique, as it partially breaks down proteins before consumption, improving digestibility.
Individual Factors
Personal factors like age and gut health also influence protein absorption. Older adults may retain more amino acids in their gut and liver tissues, reducing the amount that reaches the bloodstream. During the "first pass" through the digestive system, the gut and liver (known as splanchnic tissues) extract between 29% and 60% of absorbed amino acids for their own maintenance and enzyme production. While this is a normal process, conditions affecting gut health or enzyme production can further reduce protein absorption efficiency.
Animal-Based vs. Plant-Based Protein: Bioavailability Comparison
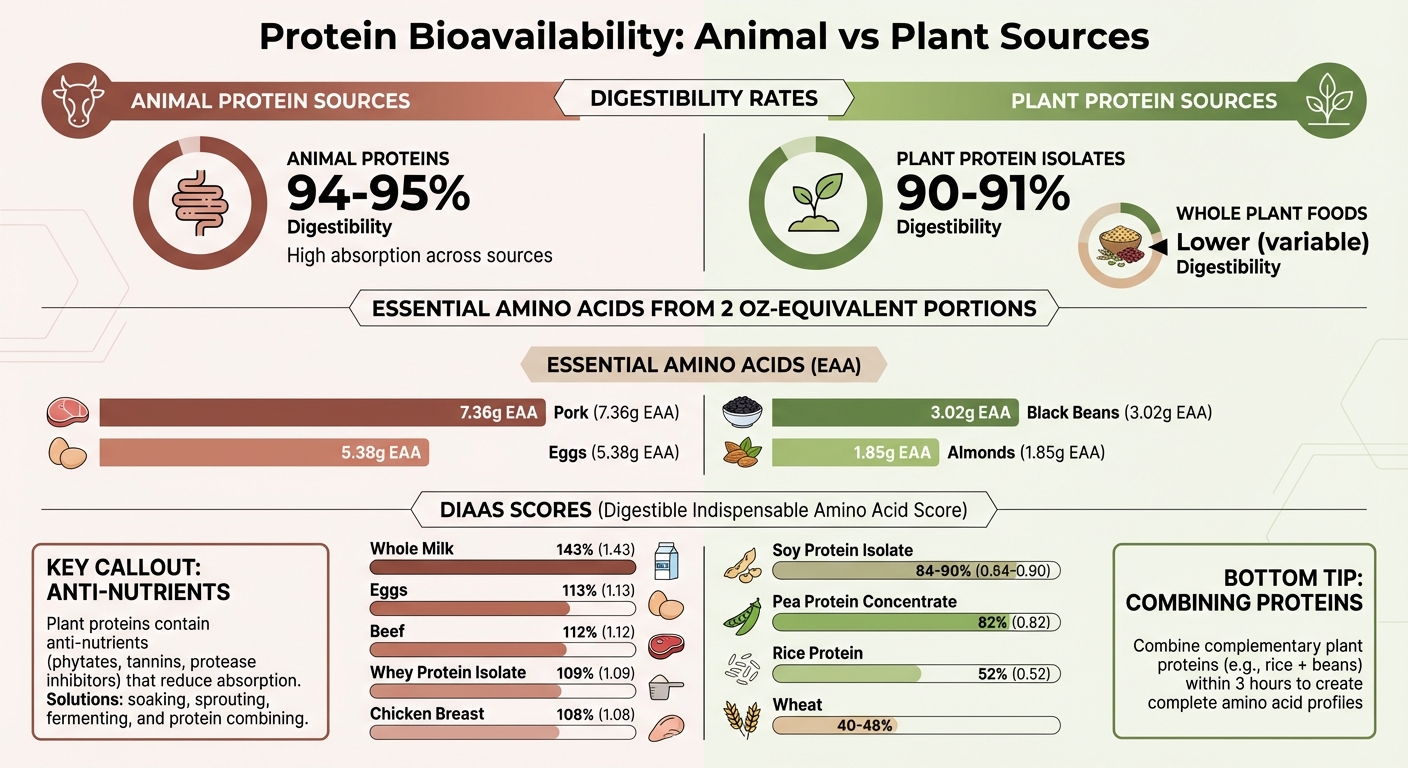
Protein Bioavailability Comparison: Animal vs Plant-Based Sources with DIAAS Scores
When it comes to protein absorption, the type of protein you consume - animal-based or plant-based - plays a huge role in how much your body can actually use. These two sources differ in digestibility, amino acid content, and the presence of compounds that can hinder absorption.
Bioavailability Rates and Digestibility
Animal proteins generally score higher in digestibility compared to plant proteins. For instance, milk proteins boast about 95% digestibility, while meat proteins reach close to 94%. On the other hand, even processed plant protein isolates like soy and pea hover around 90-91%. Whole plant foods tend to score even lower.
The differences become even more striking when looking at essential amino acids (EAAs). Research from Purdue University showed that two-ounce-equivalent servings of pork delivered 7.36 grams of EAAs, while the same portion of almonds provided just 1.85 grams - less than a quarter of pork's EAA content. Black beans landed in the middle with 3.02 grams, and eggs offered 5.38 grams.
Here’s a key takeaway: a serving of animal protein is not nutritionally equal to the same serving of plant protein. The standard serving sizes can be misleading when it comes to what your body can actually digest and use.
Here’s a quick comparison of digestibility and EAA content for common proteins:
| Protein Source | Digestibility | EAA from 2 oz-eq Portion |
|---|---|---|
| Milk | ~95% | High |
| Meat (Pork) | ~94% | 7.36 g |
| Eggs | High | 5.38 g |
| Soy/Pea Isolates | 90-91% | Moderate |
| Black Beans | Variable | 3.02 g |
| Almonds | Lower | 1.85 g |
Anti-Nutritional Factors in Plant Proteins
Plant proteins come with a catch - they often contain compounds like phytates, tannins, protease inhibitors, lectins, and saponins that can interfere with nutrient absorption.
- Phytates bind to minerals like zinc, iron, magnesium, and calcium, making them harder for your body to absorb. They also block protein-digesting enzymes.
- Tannins reduce the availability of amino acids by forming complexes with proteins and inactivating digestive enzymes.
- Protease inhibitors, found in legumes like soybeans, hinder enzymes like trypsin and chymotrypsin, which are crucial for breaking down proteins.
- Lectins can damage the intestinal lining, while saponins inhibit key digestive enzymes like amylase and lipase.
Luckily, methods like soaking, sprouting, fermenting, and cooking can reduce these anti-nutrients significantly. For grains like wheat and rice, removing the bran layer can help eliminate a large portion of phytates and tannins.
Practical Applications for Different Diets
If you consume animal proteins, you’re getting a complete amino acid profile without the complications of anti-nutrients. For example, milk protein concentrate scores 120% on the DIAAS scale, and whey protein concentrate scores 107%. These proteins require no special preparation or pairing to maximize their benefits.
For plant-based diets, however, careful planning is key. Soy protein isolate scores 84% on the DIAAS scale, while pea protein concentrate scores 62%. To create a complete amino acid profile similar to whey, you can combine complementary plant proteins. A 70:30 blend of pea and rice protein works well - rice is low in lysine but adequate in methionine, while pea and soy are low in methionine but higher in lysine.
"Fecal measurements overestimate protein digestibility because nitrogen that is transferred to ammonia by microbiota is partly absorbed in the colon".
This is why protein isolates often outperform whole plant foods in digestibility - the processing removes much of the fiber and anti-nutrients that interfere with absorption. If you’re strictly plant-based, you may need to increase your protein intake beyond the standard 0.83 grams per kilogram of body weight per day to make up for lower bioavailability. A plant-based diet typically achieves about 70% protein utilization. Tailoring your protein choices and preparation methods can make all the difference in maximizing what your body can absorb and use.
How Protein Quality Is Measured: DIAAS and PDCAAS
Protein quality is assessed using two main methods: PDCAAS (Protein Digestibility Corrected Amino Acid Score) and DIAAS (Digestible Indispensable Amino Acid Score). Since March 2013, the FAO has recommended DIAAS over PDCAAS because it measures protein absorption at the small intestine rather than relying on fecal digestibility. This shift helps us better understand how to maximize protein absorption.
The main difference between these methods lies in where and how absorption is measured. PDCAAS uses fecal digestibility and applies a single, broad digestibility value to all amino acids. In contrast, DIAAS evaluates digestibility at the ileum (the end of the small intestine) and examines each essential amino acid individually. This matters because unabsorbed nitrogen in the large intestine is consumed by bacteria, leading PDCAAS to overestimate amino acid absorption.
What Is DIAAS and Why It Matters
DIAAS takes a detailed approach, treating each essential amino acid as a separate nutrient and measuring how much of each is absorbed at the small intestine. This method reflects the body's need for specific amounts of the nine essential amino acids. The limiting amino acid - the one present in the smallest amount relative to the body’s requirements - determines the overall DIAAS score. For example, wheat scores around 40–48% on DIAAS due to its low lysine content, while whole milk powder scores as high as 116% because of its rich amino acid profile.
Unlike PDCAAS, DIAAS scores can exceed 100%. This is especially useful when combining proteins in a meal. For instance, a high-quality protein like milk (116%) can complement a lower-scoring one like wheat (40–48%), resulting in a more accurate assessment of the meal's overall protein quality. PDCAAS, however, caps scores at 100%, which hides these complementary effects.
"DIAAS is currently the most accurate score for routinely assessing the protein quality rating of single source proteins." – Paul J. Moughan, Distinguished Professor, Riddet Institute
DIAAS also uses growing pigs as a model for measuring digestibility, as their digestive systems closely resemble those of humans. Additionally, it incorporates three age-specific reference patterns (0–6 months, 6 months to 3 years, and over 3 years) instead of a single standard, making its results more precise.
While DIAAS offers a clearer picture of protein quality, understanding PDCAAS's limitations is still important for grasping the broader context of protein evaluation. Let’s explore these limitations further.
Limitations of PDCAAS
PDCAAS has several shortcomings, starting with its reliance on fecal samples to measure protein digestibility after food has passed through the entire digestive tract, including the colon. It also uses a single digestibility value for all amino acids, ignoring variations in how individual amino acids are absorbed.
Another issue is the capping of scores at 100%, which obscures differences in protein quality. For example, PDCAAS rates milk, eggs, beef, and soy protein isolate similarly - around 1.00. However, DIAAS distinguishes these proteins more effectively, giving whole milk powder a score of 1.16, beef 1.12, and soy protein isolate between 0.84 and 0.90. Furthermore, PDCAAS doesn’t account for heat damage to amino acids during cooking. Processes like the Maillard reaction, which browns food, can make lysine biologically unavailable. DIAAS, on the other hand, measures "reactive lysine", offering a more accurate assessment.
| Feature | PDCAAS (Protein Digestibility Corrected Amino Acid Score) | DIAAS (Digestible Indispensable Amino Acid Score) |
|---|---|---|
| Digestibility Site | Fecal (total digestive tract) | Ileal (end of the small intestine) |
| Truncation | Capped at 100% | Not truncated (can exceed 100%) |
| Amino Acid Focus | Single crude protein value | Individual digestibility for each amino acid |
| Animal Model | Primarily rats | Preferably pigs or humans |
| Accuracy | Generally overestimates absorption | Reflects amino acid uptake more accurately |
When researchers re-evaluated dietary data from India and Sub-Saharan Africa using DIAAS instead of PDCAAS, they uncovered protein deficiencies that PDCAAS had overlooked. This illustrates how DIAAS can guide better protein choices to meet your body’s amino acid requirements.
How to Optimize Protein Absorption
Understanding protein quality metrics is just the start. To get the most out of the proteins you consume, practical strategies can make a significant difference. Here’s how you can improve protein absorption and make your diet work harder for you.
Pick Proteins with High Bioavailability
Focus on proteins with high DIAAS (Digestible Indispensable Amino Acid Score). Animal-based proteins are among the best for muscle building and recovery. For instance:
- Whole milk scores 1.43.
- Eggs come in at 1.13.
- Beef scores 1.116.
- Whey protein isolate scores 1.09.
Cooking methods also play a role. Cooked eggs, for example, result in a 20% higher essential amino acid peak in your bloodstream compared to raw eggs. For lean meats like chicken breast (DIAAS 1.08), cooking thoroughly helps denature proteins, making them easier to digest. However, avoid over-charring, as excessive heat can cause Maillard reactions, which reduce lysine availability.
Looking for easy, high-quality options? Websites like High Protein Snacks Pro offer detailed nutritional info and DIAAS scores for various snacks, helping you make informed choices.
When high-bioavailability proteins aren’t accessible, combining different sources can help.
Pair Complementary Proteins
Plant-based proteins often have lower DIAAS scores because they are missing one or more essential amino acids. For example:
- Rice protein scores 0.52 due to low lysine levels.
- Pea protein concentrate scores 0.82 because it lacks sufficient sulfur-containing amino acids like methionine.
By pairing grains and legumes, you can create a complete amino acid profile. For instance, rice (rich in methionine but low in lysine) combined with beans (high in lysine but low in methionine) provides a balanced profile similar to animal proteins. Consuming these complementary proteins within about three hours allows your body to pool the amino acids effectively.
Time Your Protein Intake Strategically
When you eat protein matters just as much as what you eat. After exercise, your body is primed to repair muscle, making protein consumption within a few hours post-workout especially effective. This ensures amino acids are available when your muscles need them most.
Fast-digesting proteins like whey are particularly useful here. Unlike other proteins, whey doesn’t coagulate in the stomach, allowing it to empty quickly and deliver amino acids faster. For plant-based diets, spreading protein intake across meals and combining sources within a three-hour window can optimize absorption.
Prepare Proteins for Better Digestibility
Plant proteins often contain anti-nutrients - like phytic acid, tannins, and trypsin inhibitors - that hinder digestibility. Techniques such as soaking, sprouting, and fermenting can reduce these anti-nutrients and improve the protein’s bioavailability.
Cooking also plays a critical role. Moderate heat (around 194°F for 10 minutes, as used in yogurt production) helps denature proteins, making them easier for enzymes to break down. However, excessive heat can cause glycation, which significantly impacts lysine availability. For example:
- At 20% lysine glycation, plasma lysine availability drops by 35%.
- At 50% glycation, availability plummets by 92%.
To avoid this, cook meats thoroughly but steer clear of blackened or heavily charred surfaces, as these indicate advanced glycation damage.
Conclusion
Protein bioavailability plays a key role in how your body absorbs and utilizes different protein sources. Animal-based proteins like milk, eggs, and beef tend to be more efficiently absorbed due to their complete amino acid profiles and higher digestibility. For those on plant-based diets, combining complementary proteins - such as rice and beans - and using preparation techniques like soaking or fermenting can help ensure a balanced amino acid intake.
Athletes often require between 1.2–2.2 g/kg of protein daily, while older adults may need 1.0–1.2 g/kg. Prioritizing high-DIAAS (Digestible Indispensable Amino Acid Score) proteins and avoiding cooking methods like charring can significantly improve protein absorption and utilization. Timing your protein intake strategically, such as around workouts, can further enhance its benefits.
High-DIAAS proteins, including whole milk, eggs, and whey, are excellent choices due to their superior bioavailability. Additionally, using moderate heat during cooking helps retain digestibility and amino acid availability.
For a handpicked selection of protein-rich snacks with detailed nutritional information, check out High Protein Snacks Pro. Whether your goal is muscle building, weight management, or general health, focusing on proteins with better bioavailability ensures you get the most out of your intake.
Use these tips to choose, prepare, and time your protein consumption for optimal results.
FAQs
How do I choose a high-bioavailability protein?
To choose a protein with high bioavailability, focus on its quality by checking metrics such as the digestible indispensable amino acid score (DIAAS) or the protein digestibility corrected amino acid score (PDCAAS). Opt for proteins that offer a high concentration of amino acids, are easy to digest, and are efficiently absorbed by the body. These qualities help ensure your body can make the most of the protein you consume.
How can I make plant protein more complete?
Plant proteins can sometimes fall short in providing all the essential amino acids your body needs. For example, some may not have enough valine or isoleucine. The good news? You can fix this by combining different plant-based foods.
Pairing legumes (like beans or lentils) with grains (such as rice or quinoa) is a simple way to create a balanced amino acid profile. This combination fills in the gaps, ensuring your body gets the full range of essential nutrients it needs to thrive. It's an easy and effective strategy for anyone following a plant-based diet.
Does cooking change protein absorption?
Cooking does influence how our body absorbs proteins by altering their physical, chemical, and structural characteristics. These changes can either improve or reduce their digestibility and availability, making certain proteins easier - or harder - for the body to process.