Heat Processing and Protein Bioavailability

Heat Processing and Protein Bioavailability
Heat processing can improve or reduce the quality of proteins depending on how it's applied. Moderate heat unfolds proteins, making them easier for your body to digest. However, excessive heat can cause structural changes like aggregation, oxidation, and cross-linking, which decrease digestibility and nutrient availability. Key factors include temperature, duration, and storage conditions.
Key Takeaways:
- Moderate Heat: Unfolds proteins, improving enzyme access.
- Excessive Heat: Causes damage, reducing digestibility by up to 20% in some foods.
- Methods:
- Boiling/Steaming: Improves digestibility, especially for legumes.
- Baking/Extrusion: Reduces antinutrients but may lower amino acid content.
- Sterilization: Extends shelf life but can impair protein quality.
- Storage: Oxygen exposure and improper packaging can degrade proteins further.
To retain protein quality, manufacturers use techniques like controlled heating, amino acid fortification, and protective packaging. Choosing snacks processed with care ensures the protein you consume is effective for your body.
The Protein Denaturation: When Heat Defies the Rules of Cooking
sbb-itb-a09e3e8
How Heat Changes Protein Structure
Heat triggers biochemical changes in proteins that can either improve or impair their nutritional value. To understand how this works, we need to look at three key processes: denaturation, oxidation, and Maillard reactions. Each of these mechanisms affects protein structure and digestibility in unique ways.
Protein Denaturation and Digestibility
Denaturation happens when a protein's three-dimensional structure is altered, preventing it from performing its original biological role. When exposed to heat above 122°F (50°C), the hydrogen bonds and dispersion forces holding the protein's structure together break, causing the protein to unfold.
"A denatured protein cannot do its job." - Chemistry LibreTexts
This unfolding can actually improve digestibility at moderate temperatures by making the protein more accessible to digestive enzymes. But there's a catch - too much heat can have the opposite effect. For example, freeze-dried muscle protein is nearly 100% digestible, but autoclaving it can drop digestibility to just 57% to 65%.
High-heat methods like roasting and stewing can cause proteins to aggregate, oxidize, and form cross-links, including disulfide bonds. These changes make proteins harder for digestive enzymes to break down. Myofibrillar proteins, which make up 60–70% of muscle protein, are especially prone to these effects.
So, while moderate heat can help enzymes do their job, excessive heat leads to structural changes that reduce protein quality.
Protein Oxidation and Nutrient Loss
Heat-induced oxidation is another process that harms protein quality. It not only reduces the availability of essential amino acids but also makes proteins less digestible by altering their structure.
"Protein oxidation may also cause severe deterioration of nutritional value owing to the loss of essential amino acids and resistance of the oxidized protein molecules to the hydrolytic action of digestive enzymes." - Nawaz et al., Food Research International
A key sign of oxidation is the formation of protein carbonyls, which are breakdown products like aldehydes and ketones. Specific examples include alpha-aminoadipic semialdehyde (from lysine) and gamma-glutamic semialdehyde (from arginine and proline). In fact, alpha-aminoadipic semialdehyde can make up as much as 70% of total protein carbonyls in food systems.
Oxidation also causes proteins to unfold, aggregate, and form covalent cross-links like dityrosine and disulfides, leading to insolubility. For instance, when bovine plasma albumin is heated to 313°F (145°C), nearly all amino acids are significantly reduced, except for glutamic acid and those with paraffin side-chains. These structural changes affect the protein's ability to gel, emulsify, foam, and retain water, further reducing its digestibility and bioavailability.
Maillard Reactions and Lysine Loss
The Maillard reaction is a chemical process that occurs when the free amino groups in proteins react with the carbonyl groups in reducing sugars. While this reaction adds appealing flavors and colors to cooked foods, it comes at a nutritional cost.
Lysine, along with arginine, histidine, and tryptophan, is particularly vulnerable to the Maillard reaction. When lysine is modified into compounds like Amadori products or Advanced Glycation End-products (AGEs), it becomes nutritionally unavailable, meaning your body can no longer use it as an essential amino acid.
"If lysine is modified as an Amadori product via the Maillard reaction, the nutritional value of the protein is significantly reduced because the modified lysine can no longer be utilized by the human body." - Ines Kutzli, Department of Food Material Science, University of Hohenheim
This is especially concerning because lysine is often the limiting amino acid in plant-based proteins. The glycation process also blocks the sites where digestive enzymes like trypsin and chymotrypsin would typically cleave the protein, further reducing digestibility. In studies, diets high in Maillard reaction products have shown 47% higher fecal nitrogen excretion and 12% lower nitrogen absorption, highlighting the nutritional drawbacks of this process.
While the Maillard reaction enhances flavor and appearance, it compromises the availability of lysine and other essential amino acids, directly impacting the protein's nutritional profile.
How Different Heat Processing Methods Affect Protein Quality
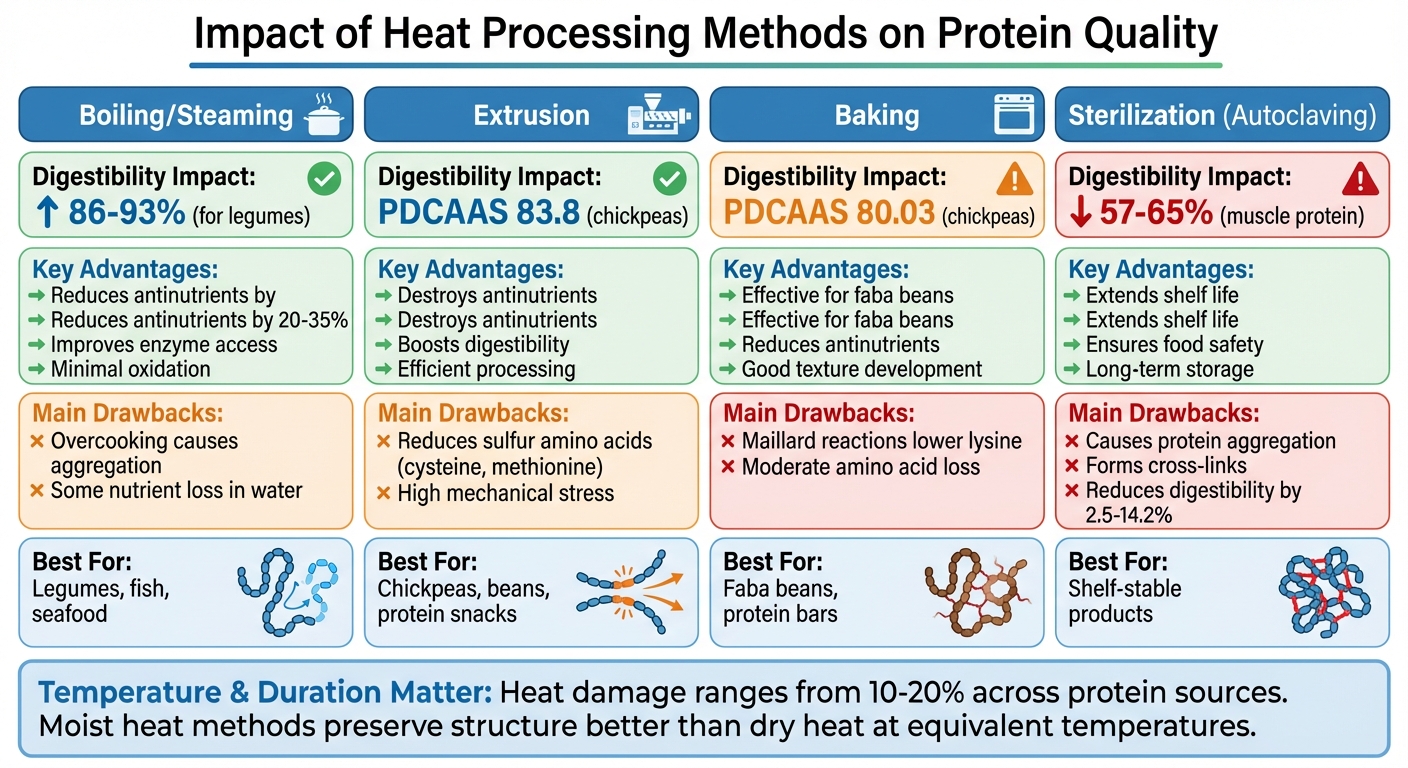
How Heat Processing Methods Affect Protein Digestibility and Quality
Different heat processing methods can significantly influence protein quality. Choosing the right technique is key to preserving nutrient availability, especially in high-protein snacks. Let’s break down how these methods impact protein integrity.
Boiling and Steaming
Boiling and steaming, both moist-heat methods, generally enhance protein digestibility. They work by unfolding proteins, making them more accessible to digestive enzymes. These methods are particularly effective for plant-based proteins, such as legumes, which contain antinutrients that can block protein absorption.
Research published in Food Chemistry shows that boiling can increase legume digestibility from 33–38% to 86–93%, while reducing antinutrients by 20–35%. Cooking at 250°F (121°C) for 10 minutes is ideal for digestibility, but extending cooking times can lead to protein aggregation, making digestion harder.
"Thermal processes such as sous-vide can induce favorable changes, such as partial unfolding or exposure of cleavage sites... whereas processes such as stewing and roasting can induce unfavorable changes, such as protein aggregation." - Institute of Food Technologists
For muscle proteins, like fish and seafood, steaming is a better choice than roasting or frying. Unlike high-heat methods, steaming minimizes oxidation and the formation of disulfide bonds, which can impair digestion. The key takeaway? Use moist heat to cook food until just tender, avoiding overcooking to maintain protein quality.
Baking and Extrusion
Dry-heat methods like baking and extrusion are widely used in food processing. These techniques effectively destroy antinutrients, such as trypsin inhibitors, but they can also damage protein structure if temperatures get too high.
Extrusion combines high heat, pressure, and mechanical forces to unfold proteins, improving their digestibility. For example, extruded chickpea flour achieved a Protein Digestibility Corrected Amino Acid Score (PDCAAS) of 83.8, compared to 80.03 for baked chickpea flour. Similarly, in Phaseolus bean varieties (e.g., black, navy, pinto, and red kidney), extrusion increased digestibility to an average PDCAAS of 66%, compared to 52% for baked beans.
However, extrusion comes with trade-offs. While it boosts digestibility, it reduces sulfur amino acids like cysteine and methionine due to the high temperatures and mechanical forces involved.
"Extrusion does not alter protein content in beans, but has been shown to reduce the content of both cysteine and methionine, potentially due to the disruptive forces of the extruding process as well as the high temperatures used in extrusion." - Matthew G. Nosworthy, University of Manitoba
Interestingly, the effectiveness of these methods varies by crop. For instance, baked faba beans have shown higher digestibility scores (PDCAAS 66%, DIAAS 61%) than their extruded counterparts. This highlights the importance of tailoring processing methods to specific protein sources.
High-Temperature Sterilization
High-temperature sterilization, such as autoclaving or retorting, is essential for extending shelf life and ensuring food safety. However, it can significantly impair protein bioavailability if not carefully managed.
Sterilization damages proteins through aggregation, oxidation, and the formation of covalent cross-links like disulfide bonds. These changes make proteins harder for digestive enzymes to break down. For example, autoclaving can lower protein digestibility in sunflower seed meal, cottonseed flour, and corn by 2.5 to 14.2 percentage points. The Maillard reaction, which occurs at high temperatures, further reduces the availability of lysine, an essential amino acid.
"The challenge to the food processing industry is to minimize the loss of nutrients during thermal processing while providing an adequate process to ensure an extended storage life." - Springer Nature
To mitigate these effects, manufacturers can use High-Temperature Short-Time (HTST) or aseptic processing, which apply heat more efficiently to preserve nutrients. Controlling moisture levels during and after sterilization is also crucial to prevent further nutrient loss during storage.
| Processing Method | Impact on Digestibility | Key Advantages | Main Drawbacks |
|---|---|---|---|
| Boiling/Steaming | Increases legume digestibility to 86–93% | Reduces antinutrients; improves enzyme access | Overcooking leads to aggregation; nutrient loss |
| Extrusion | PDCAAS of 83.8 for chickpeas | Destroys antinutrients; boosts digestibility | Reduces sulfur amino acids |
| Baking | PDCAAS of 80.03 for chickpeas | Effective for faba beans; reduces antinutrients | Maillard reactions lower lysine levels |
| Sterilization | Reduces digestibility in some proteins | Extends shelf life; ensures safety | Causes aggregation and cross-linking |
Moist-heat methods like boiling and steaming are gentler on protein structure, while dry-heat methods such as baking and extrusion excel at reducing antinutrients but may compromise amino acid content. Sterilization, though critical for shelf-stable products, requires precise controls to minimize nutrient loss.
Factors That Affect Protein Bioavailability During Heat Processing
When it comes to maintaining protein quality during heat treatment, understanding the key influences can help both manufacturers and consumers make informed decisions. Two critical aspects to consider are the temperature and duration of heat exposure and the conditions during storage.
Temperature and Duration
The balance between enhancing and damaging protein quality during heat processing depends largely on the temperature and how long the heat is applied. Gentle heating can improve digestibility by unfolding proteins, which exposes enzyme sites. But too much heat or prolonged exposure can lead to protein aggregation, oxidation, and cross-linking, ultimately reducing quality.
Interestingly, the impact varies by food type. For instance, high-temperature commercial processing has been shown to lower protein digestibility in sunflower seed meal, cottonseed flour, and corn by 2.5 to 14.2 percentage points. Across other protein sources, total heat-related damage can range from 10% to 20%, depending on the specific food.
"Protein oxidation (POx) is a process that leads to conformational and structural alterations, aggregation, and fragmentation, which consequently impairs the functional properties of proteins and thus affects meat quality." - Annual Review of Food Science and Technology
Moist heat has a particularly strong effect. For example, soybeans processed at 250°F (121°C) for one hour experience significant changes, such as reduced soluble protein and lower digestibility. Dry heat at the same temperature, however, has a much milder impact. Beyond the heat treatment itself, the conditions during storage also play a big role in protein quality.
Moisture and Storage Conditions
Storage conditions after heat processing are just as important. Proteins continue to degrade over time due to oxidation, chemical reactions, and interactions with lipids.
Exposure to oxygen accelerates this process. High-oxygen packaging, for instance, promotes oxidative damage to muscle proteins, reducing their nutritional value. Even freezing foods at -4°F (-20°C) doesn’t completely stop these changes. Research on silver carp surimi has shown that after 90 days in the freezer, significant oxidation occurs in myofibrillar proteins, marked by increased carbonyl content and structural damage.
"Nutrients can undergo unwanted degradation upon processing and subsequent storage, negatively influencing the nutritional value of food and its physiological effects." - Vibeke Orlien and Tomas Bolumar
To preserve protein quality, the storage environment must be carefully controlled. Vacuum packaging or modified atmosphere packaging with reduced oxygen levels can dramatically slow protein degradation. High-carbon-dioxide packaging, for example, has been particularly effective in preventing myofibril breakdown and oxidation in refrigerated fish fillets compared to standard air packaging. Consistent refrigeration and the use of protective additives, like antioxidants, can further stabilize protein during storage. These strategies offer practical ways to protect protein integrity in heat-processed foods.
How to Retain Protein Quality in Heat-Processed Snacks
Heat processing can pose challenges to maintaining protein quality, but manufacturers have tools at their disposal to address these issues. By carefully managing heat exposure and incorporating strategic fortification methods, it's possible to create snacks that offer both great taste and nutritional value.
Minimizing Heat Exposure
Preserving protein integrity often starts with reducing heat exposure. Using the shortest cooking time at the lowest effective temperature is key. For liquid protein bases, direct steam heating offers an effective solution. Research from 10 plants across six countries revealed that milk treated with direct steam heating experienced β-Lactoglobulin denaturation levels between 35–80%. In contrast, indirect steam heating caused significantly higher denaturation rates, ranging from 79–100%. The rapid heating and cooling in direct steam systems, which avoids direct contact with heat transfer surfaces, plays a critical role in limiting protein damage.
For solid snacks, steaming and microwaving are effective methods for reducing water exposure and shortening cooking times, which helps retain soluble nutrients. Extrusion, when carefully controlled, can also ensure high protein quality. Standard HTST pasteurization, performed at 161.6°F (72°C) for 15 seconds, results in minimal whey protein denaturation. These examples highlight the importance of precise temperature management to protect protein quality during processing.
Fortifying with Amino Acids
In addition to managing heat exposure, amino acid fortification is a powerful strategy to restore nutritional value. High-temperature processing can reduce the digestibility of certain amino acids, particularly lysine. Post-processing fortification helps maintain the Protein Digestibility-Corrected Amino Acid Score (PDCAAS) by replenishing these essential nutrients.
Fermentation, often used to enhance texture, requires careful monitoring of sulfur-containing amino acids, as some bacterial strains can break them down during the process. By addressing these potential losses, manufacturers can ensure their products remain nutritionally robust.
Product Examples from High Protein Snacks Pro
Several products featured on High Protein Snacks Pro showcase how effective strategies can preserve protein quality. For instance, Quest Nutrition Protein Bars deliver 20 grams of protein per serving. Their formulation minimizes conditions that promote the Maillard reaction, which can degrade protein quality. Two Good Greek Yogurt offers 12 grams of protein in an 80-calorie serving, using gentle pasteurization techniques to protect whey protein structure and bioavailability. Similarly, Quest Nutrition Protein Chips provide 19 grams of protein per serving through a baking process optimized for nutrient retention while achieving the desired texture.
These examples highlight how careful processing techniques and thoughtful formulations can result in snacks that deliver both high protein content and excellent nutritional quality.
Conclusion
Heat processing plays a dual role in protein nutrition. On one hand, it ensures food safety and extends shelf life. On the other, it can strip away important nutrients if not done carefully. The difference between protein that fuels your fitness goals and protein that falls short lies in how manufacturers manage their heat processing techniques.
Gentler methods like sous-vide or steaming can actually enhance digestibility by unfolding proteins and exposing enzyme cleavage sites. In contrast, high-heat techniques like roasting or stewing often lead to protein aggregation and cross-linking, which make digestion more difficult. Research shows that intense heat can have a dramatic impact - autoclaving muscle protein, for example, reduces its digestibility from nearly 100% to just 57–65%. Plant-based proteins aren't immune either, with digestibility dropping by 2.5 to 14.2 percentage units in foods like sunflower seed meal and corn.
"The challenge to the food processing industry is to minimize the loss of nutrients during thermal processing while providing an adequate process to ensure an extended storage life." - Springer Nature
These insights highlight the importance of choosing snacks that preserve protein bioavailability. Just because a label says "20 grams of protein" doesn’t mean your body will absorb all of it. The real nutritional value depends on how well the processing methods maintained the protein's quality. High Protein Snacks Pro helps you navigate these details by offering clear nutritional breakdowns and spotlighting products that use controlled heat processing to retain protein integrity.
Whether you’re grabbing a protein bar, chips, or Greek yogurt, understanding how heat impacts protein bioavailability can help you make smarter choices. The right processing methods ensure that the protein you consume actually delivers the benefits your body needs.
FAQs
Does cooking protein always make it easier to digest?
Cooking protein doesn’t always make it easier to digest. Heat can change the protein's structure and chemical makeup, which might lower its bioavailability and make it harder for the body to process. How much this happens depends largely on the cooking method and the temperature applied.
Which cooking methods best preserve amino acids like lysine?
Moist heat cooking methods, whether in open or closed vessels, are better at preserving lysine, retaining about 69–83% of this essential amino acid. On the other hand, dry heat methods, such as microwaving or using a hot air oven, cause more lysine loss, with retention dropping to 31–65%. To keep more lysine in your food, stick with moist cooking techniques whenever you can.
How can I tell if a high-protein snack was over-processed?
Over-processed high-protein snacks often reveal their nature through noticeable changes in texture, flavor, or appearance, which result from excessive heat, pressure, or chemical treatments during manufacturing. These aggressive processes can break down proteins, making them less digestible or reducing the availability of essential amino acids. In some cases, harmful compounds may even form as a byproduct.
To spot signs of over-processing, pay attention to the detailed nutritional information on packaging. Reduced protein quality or digestibility can be red flags. Understanding how the product was processed can also provide valuable insight into its overall nutritional value.